The US and France are the only other countries to have formally approved pre-exposure prophylaxis, a pull used by HIV-ngeative gay men as a form of HIV prevention.
Canada has followed in the footsteps of the US and France by becoming the latest country to approve HIV-prevention medication pre-exposure prophylaxis (PrEP).
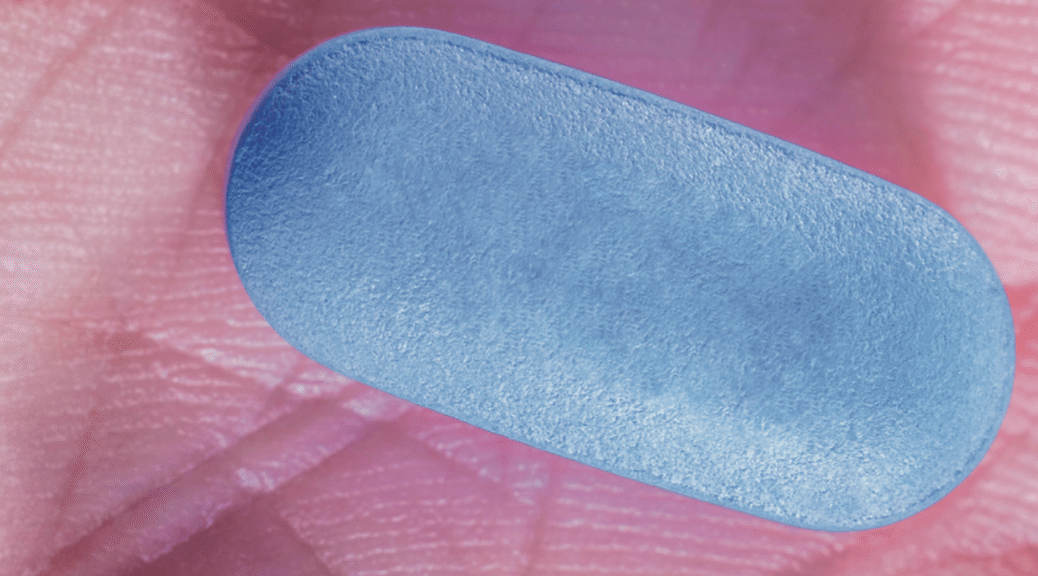
Canada’s national department of health, Health Canada, formally approved PrEP on February 23 after Gilead — the maker of the PrEP drug Truvada — submitted an application for approval in 2015
“The drug is intended for use by high risk individuals – such as those whose sexual partner is HIV-positive – in combination with safer sex practises, including condom use,” a Health Canada spokesperson told BuzzFeed Canada.
The approval of PrEP is expected to lead to lower prices for the drug in Canada, as well as a potential increase in prescriptions. The US has had PrEP since 2012 while France only had it approved last November. The World Health Organisation has also endorsed it. In Australia, doctors can prescribe the medication to patients but it is only available to those who can afford to import it from online pharmacies.
Truvada is currently awaiting approval from the Therapeutic Goods Administration (TGA) and from there it must still be approved to go onto the Pharmaceutical Benefits Scheme (PBS) to make it affordable for most people. The TGA recently pushed its deadline to make a decision on PrEP to May this year.
Like Australia, the UK is also facing pressure to approve PrEP.
Following Canada’s approval of the drug, the Greens last week on the Federal Government to fast-track its approval in Australia.
“There is now a strong evidence base and consensus amongst Australian non-government organisations working in this area that PrEP, along with rapid HIV tests and home self-tests are vital to add to the prevention tools currently available, so the government should be doing all it can to speed up availability,” South Australian Greens Senator Robert Simms said.
“Australia is lagging behind.”
Last November, the Senate backed Simms’ motion calling on the Australian Government to remove regulatory barriers to access to PrEP, rapid HIV tests and home self-tests, and urgently explore policy options — including expanded trials in states and territories — to enable access to these important prevention tools.
According to Ending HIV, a national campaign committed to ending HIV transmissions by 2020, PrEP is “use of antiretroviral drugs, taken by HIV negative people to prevent HIV infection”.
It also says Truvada is the only drug that has been proven to work, with “others are on the horizon”.
Ending HIV’s website also features a guide on how to obtain Truvada within Australia. Additionally, advocacy group PrEPAccessNow has assistance resources for Australians facing financial hardship in order to make the drug as accessible as possible.
Article | Star Observer